Structural Heart Disease
Frontier Explorer
STRUCTURAL HEART DISEASE
Recently, a team from the Heart Research Institute of Zhongshan Hospital affiliated with Fudan University conducted an exploratory study on 10 patients with functional mitral regurgitation (FMR) using the world's first transapical mitral valve clamp ValveClamp, developed in collaboration with Shanghai Hanyu Medical Technology Co., Ltd. (referred to as Hanyu Medical), and achieved satisfactory results. This study was conducted by Academician Ge Junbo, Director Wang Chunsheng, Professor Zhou Daxin, Professor Wei Lai, and Professor Pan Cuizhen.
The study included 10 patients with an average age of 71.0+/-8.5 years. Including 4 patients with atrial functional mitral regurgitation and 6 patients with ventricular functional mitral regurgitation. Three of them were anatomically challenging and not suitable for MitraClip surgery patients. The success rate of the surgery was 90%. One patient had unsatisfactory intraoperative results due to challenging anatomy and switched to traditional surgical procedures. Postoperative ultrasound showed that among the 9 successful patients, 7 had reflux reduced to grade 0-1 and 2 had grade 2. These two patients were anatomical challenge patients, and their postoperative symptoms also improved significantly. Seven out of nine successful patients used one clip, while two challenging cases used two clips. The most surprising thing is that the median catheter surgery operation time is 10 minutes, which means that half of the cases have a catheter surgery operation time of less than 10 minutes, with the fastest being 5 minutes.
News: A minimally invasive mitral valve repair surgery with a catheter operation time of only 5 minutes

Figure 1 shows a patient's preoperative and postoperative images
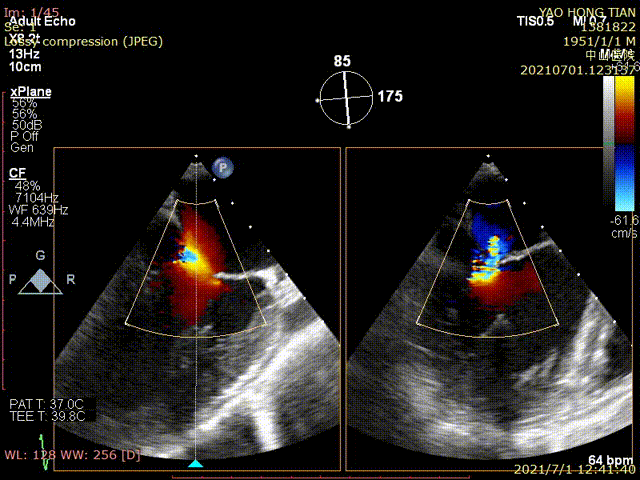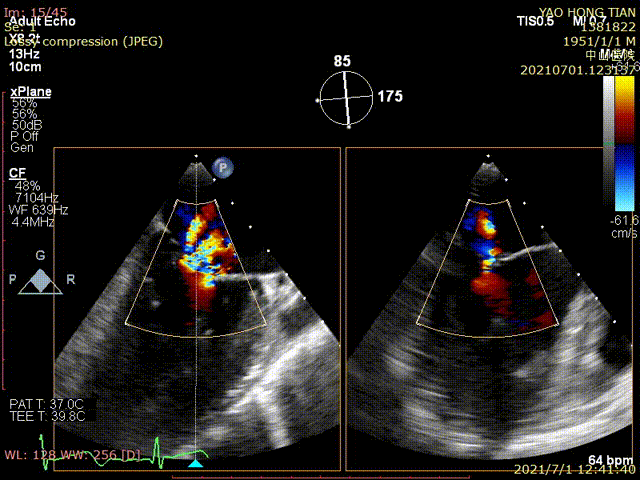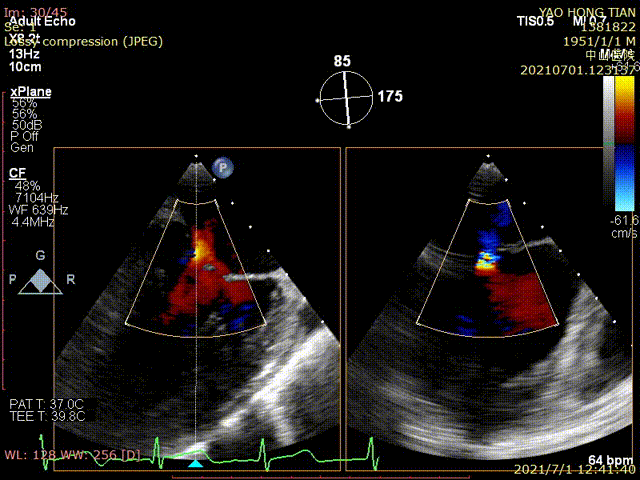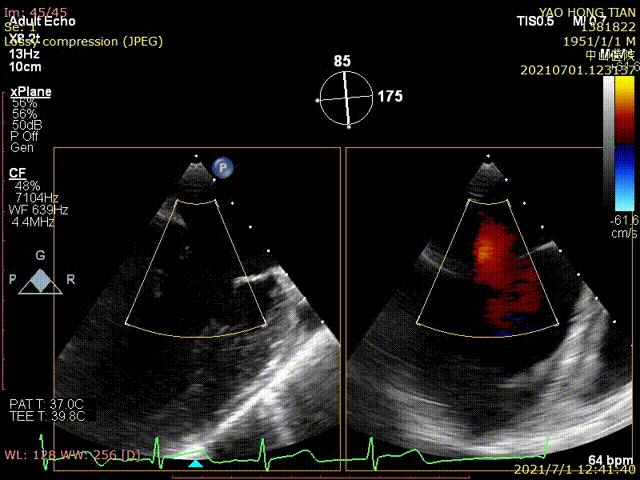
Preoperative ultrasound
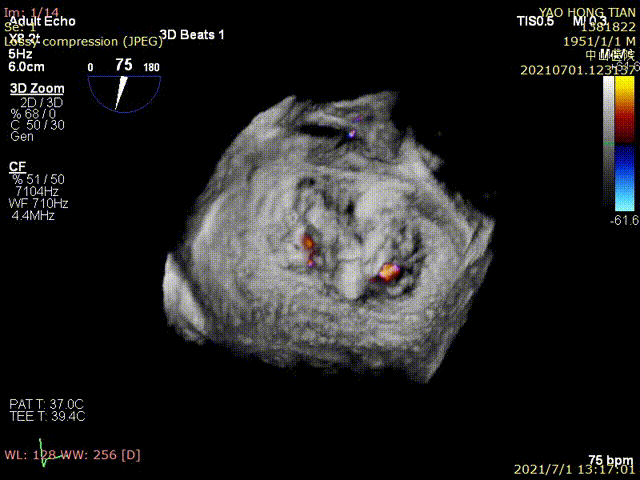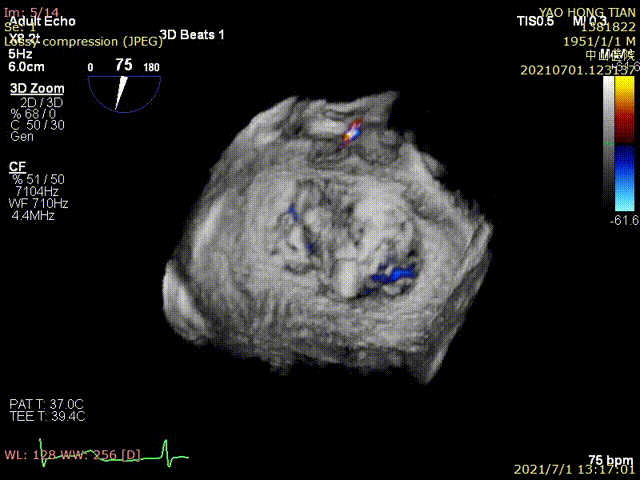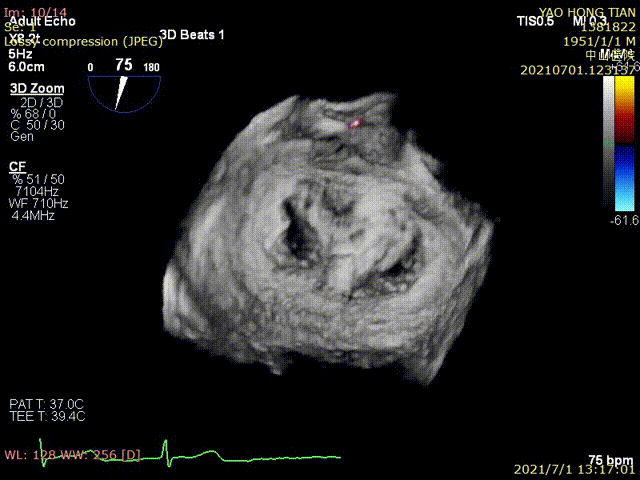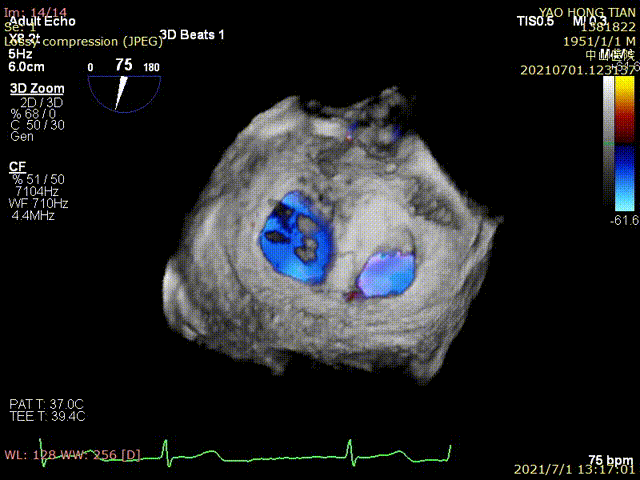
Postoperative ultrasound
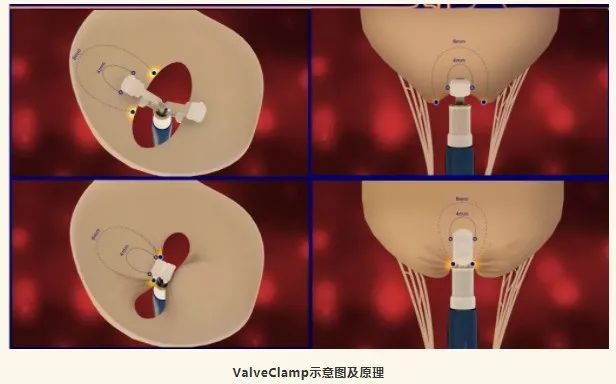
ValveClamp is the world's first transapical mitral valve clamp, which has been granted PCT patents in multiple countries worldwide. This device has some unique advantages compared to other transductal edge to edge repair (TEER) devices currently available internationally:
① The surgery time is short and the learning curve is short. Based on current research data, the ValveClamp catheter operation time is significantly shorter than similar international devices. Even at the current level of technology, the average operating time for TEER instrument catheters internationally is 1.5 hours, and in challenging cases, it often reaches 4-6 hours. ValveClamp, on average, only takes 10 minutes, which gives it a significant advantage in this regard.
② No DSA machine or catheterization room is required, it can be completed in a regular operating room. ValveClamp has low hardware requirements and is suitable for large-scale promotion. At present, the DSA machines and catheterization rooms in various hospitals have very limited resources, which is an important factor restricting the promotion of interventional surgery. ValveClamp surgery can be performed with a single ultrasound machine in general surgery (currently, intraoperative ultrasound is required for routine surgical procedures). The surgeon does not require DSA operation experience or catheter operation experience, and can easily get started, especially suitable for surgeons.
③ Low production cost, suitable for China's national conditions and national strategy. The ValveClamp has a simple structural design and a significantly simplified production process compared to similar instruments, making it possible to effectively control production costs. After going public in the future, the market price can be lower, saving costs for patients, which is in line with the current national policy trend.
Professor Wei Lai, the main operator of this study, said thatthere are many cases of functional mitral regurgitation, and the incidence rate is 3-4 times higher than that of patients with degenerative mitral regurgitation. Most of these cases cannot be operated in previous cardiac surgery. Even if these patients use foreign TEER instruments, the surgery is still very challenging. We have conducted multiple trials using ValveClamp for both atrial and ventricular FMR, and found that ValveClamp is indeed simple, fast, and effective, especially since the instrument is very easy to operate, easy to learn, and has great application prospects.
The preclinical study of ValveClamp in China, which only included primary mitral regurgitation, was completed at the beginning of this year and the patients are currently being followed up. This device is expected to be approved for market by the end of 2022, becoming the first interventional treatment device for mitral regurgitation in China.